Managing infectious hemolymphatic diseases (Proceedings)
Infectious and non-infectious hemolymphatic diseases generally present with the same clinical symptoms due to cardiovascular insult, lack of tissue oxygenation and possible impending cardiovascular collapse.
Major management issues
Clinical recognition of disease
Infectious and non-infectious hemolymphatic diseases generally present with the same clinical symptoms due to cardiovascular insult, lack of tissue oxygenation and possible impending cardiovascular collapse. Frequently, bacterial sepsis is hard to differentiate from viral diseases. Endotoxemia due to gastrointestinal infections is always a crucial rule out. When red blood cell destruction occurs with these diseases, then clinical signs of primary anemia may be the overriding clinical presentation. General clinical signs will include mucous membrane pallor, possible deposition of pigment in mucous membranes, tachycardia, tachypnea, weakness, systolic murmur,
Exercise intolerance, collapse, blindness, ileus, laminitis, and evidence of renal failure. It is important to determine if anemia is present if there is intravascular or extravascular destruction (Figure 1).
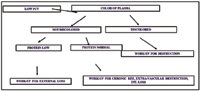
Figure 1. Algorithm of Anemia
A second issue is identification of primary vasculitis as a syndrome. Identification of vasculitis is further complicated by whether or not a cellulitis is present as opposed to a vasculitis (Table 1). Third, the clinical diagnosis is frequently confused by whether or not there is primary red blood cell destruction, platelet destruction, or both.

Table 1. Most Common Identified Infectious Causes of vasculitis and cellulitis
Equine granulocytic ehrlichiosis
Equine granulocytic ehrlichiosis caused by Anaplasma phagocytophilum (syn. Ehrlichia equi, Anaplasma equi) is a tick-borne disease characterized by fever, anorexia, depression, limb edema, jaundice, petechiation, reluctance to move, and ataxia. While the disease is most common in California, outbreaks have been reported throughout the Northern and Southeastern United States. Now that this disease is correctly recognized as A. phagocytophilum, clearly this is one of the most common causes of infectious anemia and thrombocytopenia in domestic animals. This disease is currently considered the most common tick-borne disease of animals in Europe. However there are many "genotypes" of this organism which may result in some phenotypic presentations that differ in hosts and geographically so that disease is identified in one species in a particular locale more often than another. Occurrence of disease may also be dependent on tick species which harbor the bacterium. Nonetheless, emergence of this disease can occur in a particular mammalian species or tick given the dynamic variation in gene expression of this organism. This organism has a very broad host range and actually sheep are a very important reservoir. In addition mice and deer are also reservoirs which maintain this organism within the environment.
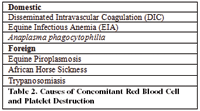
Table 2. Causes of Concomitant Red Blood Cell and Platelet Destruction
Diagnosis
In the horse, this disease is likely under diagnosed. Typical clinicopathologic changes include leukopenia, thrombocytopenia, and hyperfibrinogenemia. Anemia is temporary and mild. This organism infects granulocytes and eosinophils not platelets, and the mechanism for thrombocytopenia is unknown at this time. This is mostly a late summer, fall disease and carried by Ixodes spp. ticks. but expansion has occurred in several Ixodes spp. This is an ACUTE DISEASE: this does NOT PERSIST. Incubation is 9-11 days and clinical signs last 7-14 days. Clinical signs include Fever, anorexia, depression, limb edema, jaundice, petechiation, reluctance to move. Ataxia can be severe. Clinical disease is less severe in younger horses. Ventricular arrhythmias can occur and death can occur with this disease! Clinical Pathology is identified by leukopenia, thrombocytopenia, hyperfibrinogenemia. Intracytoplasmic inclusions can be seen in granulocytes. Once needs to examine at least three/slides.
• PCR is quite sensitive early to late: Send in Buffy Coat to Laboratory Within 48 hours of collection, send in purple top tube (3- 5 ml size best, if smaller send 2-3) for buffy coat PCR. Before send one sends in the tube, make three blood smears.
• Treatment of choice is tetracycline at 7 mg/kg IV once per day for 7 days. Do not recommend doxycycline because oral absorption is variable.
• IF NO IMPROVEMENT IN 12-24 hours think of another dz!
• If survive acute stages, recovery in 10-14 days
Equine infectious anemia virus
Equine infectious anemia virus is an important disease of horses in terms of its potential to harm valuable livestock bloodlines and quarantine important segments of the industry. EIAV is still present in the United States with sporadic outbreaks occurring throughout the states. Although considered more prevalent in the Southeast, recent outbreaks in the West indicate that the disease is problem for all horse owners. EIAV is caused by Lentivirus of the retrovirus family. The virus is non-oncogenic and infects primarily macrophages. Transmission (natural) occurs through the blood feeding of primarily Tabanus spp. (Horseflies).
Equine infectious anemia virus has always been present outside the North America, with a worldwide distribution. Infection in Central and South America is not uncommon. Outbreaks have been sporadic in Europe, with the latest outbreak of grave concern in Irish bloodstock. The source of this infection was determined to be imported plasma from Italy. The horses exposed to index case were extensively followed and tested providing excellent information regarding field testing in a new outbreak (Discussed later under Diagnosis).
Clinical disease
Three clinical syndromes can occur in horses with EIAV:
• Acute
• Chronic
• Inapparent carrier
Classically, horses develop clinical signs of EIAV around 30 days after infection and become serologically positive by 16-42 days post-infection. In acute infection (initial viremic episode), the virus invades and undergoes massive replication in macrophages. Thrombocytopenia is the most consistent finding during initial infection, leukopenia with lymphocytosis, and moncytosis also occurs. After the acute stage, chronic lifelong infection occurs. During the end of this acute cycle and subsequent cycles, PCV, RBC, and hemoglobin decrease allow with an increase in body temperature during this time. Horses may not show overt signs of disease even when cycling. Long term, clinical signs include weight loss, edema of the limbs and ventral abdomen, and cyclical fevers. Anemia and thrombocytopenia occur with fever. Horses can also develop ataxia, liver disease, and renal disease. Fatality is observed during the acute to early chronic phase. However, most horses recover and either cycle chronically or have no clinical signs and serve as infectious, inapparent carriers.
Diagnostic testing
The agar gel immunodiffusion (AGID) test historically has been the Federal regulatory test for screening and identifying EIAV infected horses and is still considered the 'gold standard' by the OIE. Recently, an ELISA has been allowed for regulatory purposes and provides for more for rapid testing methodology. In the Irish outbreak illustrates some of the conundrums involved in testing and confirming EIAV infection in horses. The AGID tended to be more reliable in clinically ill animals, with time from exposure to seropositivity longer with this technique than with the ELISA. In addition not all EIAV ELISA's (there are several commercial tests) agree consistently in reactivity during the period post-infection. However, the ELISA overall will detect horses sooner than the AGID. Identification of virus (both replicating and provirus) was the most reliable by PCR to detect subclinical infection and horses with virus before onset of clinical diseases. Although seroconversion has been document to occur within 60-90 days post exposure, some horse took up to 200 days to seroconvert in this outbreak. In addition, in several clinical horses, seroconversion did not occur until after the first cycle.
Emergence of this disease in locales is exceedingly expensive; over 25,000 samples were tested during this new outbreak. Indeed, if there is an estimated 6 M horses in the U.S., there are likely 1.5 M tests run annually. Still this only accounts for approximately ⅓ of horses in the U.S. that are tested annually.
Treatment
There is no treatment for EIAV, other than supportive should survival beyond initial infection occur. Individual states and the federal government require either humane destruction and/or quarantine of EIAV infected horses for life. The exact quarantine methods are somewhat variable from state to state, but a 200-yard distance between affected horses and a susceptible population is required. Some states also require screens or indoor housing in addition to the physical quarantine. In an outbreak situation, horses are usually quarantined at 90 days to detect post-exposure seroconversion. Retesting is done every 30 to 60 days depending on the requirements of local regulatory officials. Clearly with this latest data, horses may be best quarantined post-exposure for several months pending retesting.
Equine viral arteritis virus
• Latest outbreaks: NM, WI, MN, MI, IL.
• Common Syndromes: Respiratory, abortion, chronic shedding in semen by asymptomatic stallions
• Less commonly described: Neonatal disease characterized by severe respiratory signs and high mortality. This Togavirus infects the arteries of horses causing fibroid necrosis of the tunica media. Animals develop a secondary leukopenia and severe thrombocytopenia. Organ failure (mostly kidney can result). The virus can persist, but usually persistence is confined to stallions, being shed primarily in semen. These carrier stallions can shed for life. Sometimes, these animals can clear; once castrated the animals will clear virus. The incubation period is 3-14 days. The most common introduction is through infected semen into a clean facility full of naïve animals.
Clinical signs
In the US, most cases are subclinical!! If nonreproductive signs occur, there is fever, limb edema, rhinitis, conjunctivitis, periorbital swelling, photophobia, stiffness. Respiratory and vasculitis can persist.
Reproductive effects
In utero death occurs in 90-120 day fetuses and in foals infected in utero around 1-3 weeks after exposure, death may occur 2-4 days after birth. There is no decrease in fertility of mares after infection, although stallions may have a temporary decrease in fertility.
Diagnosis and regulatory testing
The serum neutralization test is considered the regulatory standard. A titer of >1:4 is considered positive. There is no way to distinguish antibodies derived from infection from that of vaccination.
In the light of the recent outbreaks these are the current recommendations: All stallions of unknown status should be tested for EVA prior to breeding, if seronegative and being naturally bred to mares of unknown status, vaccinated in 10 days after testing. If positive, the semen of that horse should be cultured or tested by PCR for the virus. If the virus is detected in the semen, this animals should be considered a carrier. If any mare seroconverts to a positive stallion within 21 days, this stallion should be deemed a carrier. The virus is not killed through shipment nor freezing procedures.
Vaccination guidelines
• Intact Colts: Vaccinate at 6-12 months of age before breeding
• Yearlings: Annual vaccines for colts and breeding stallions
• Pleasure and Performance Horses: Annual vaccine for colts intended to be breeding stallions
• Broodmares: Annual vaccine for seronegative, OPEN mares 28 days before breeding
• Stallions: Annual vaccination for breeding in stallions and teasers 28 days before the start of breeding
• Keep Documentation of ALL Vaccination Records for SALES, SHOWS AND INTERNATIONAL SHIPPING
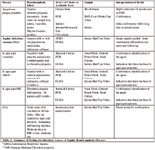
Table 3. Summary of Testing for Infectious Causes of Equine Hemolymphatic Diseases